Abiobag Singe-Use Bioreactor Bag
Abiobag Singe-Use Bioreactor Bag
Abiobag Singe-Use Bioreactor Bag
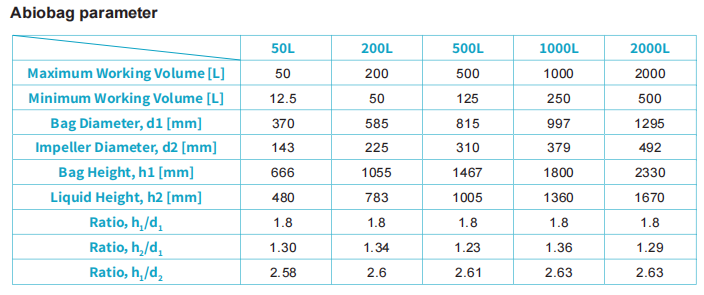
The AbioSUS single-use bioreactor system is designed based on the principles of traditional stirred-tank biore.actors, featuring a defined aspect ratio and optimized impeller positioning. This ensures high process consistency between single-use and stainless-steel systems, enabling seamless scale-up and technology transferfrom R&D to manufacturing. The Abiobag single-use bioreactor bag provides an excellent environment for cellgrowth, supporting both standard fed-batch processes and intensified perfusion cultures.
The AbioSUS single-use bioreactor system is designed based on the principles of traditional stirred-tank biore.actors, featuring a defined aspect ratio and optimized impeller positioning. This ensures high process consistency between single-use and stainless-steel systems, enabling seamless scale-up and technology transferfrom R&D to manufacturing. The Abiobag single-use bioreactor bag provides an excellent environment for cellgrowth, supporting both standard fed-batch processes and intensified perfusion cultures.
Scalable Performance
Abiobag maintains a center-mounted vertical impelle!design.consistent with conventional stirred-tank reactors. This significantly reduces the risks of variationsin cell density, viability, and product quality duringscale-up.Whetherscaling from bench-top glass biore.actors or transferring to stainless-steel systems.Abiobag ensures robust and predictable processperformance.
eFlexible Customization
Abiobag offers extensive customization optionsincluding flexible tubing configurations in terms ofsize,length,and layout, and compatibility with a widerange of probe types such as traditional reusableprobes,single-use patch sensors,and opticasensors.
Batch-to-Batch Consistency
Abiobag is manufactured using the well-establishedRenolite 9101 film, ensuring reliable and consistentperformance,and is supported by strict quality controland a comprehensive validation package that guarantees consistent batch quality, compliance with regulatory requirements,verified extractables and leachables(E&L)perfomance,and proven biocompatibility.
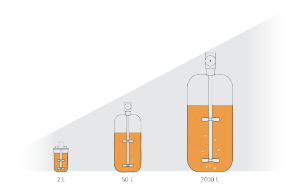
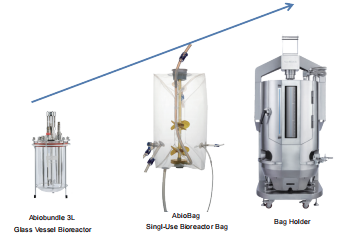
True Linear Scale-Up
Applitech offers a complete bioreactor portfolio includ.ing glass bioreactors, single-use bioreactors, and stainless-steel systems.All platforms follow a unified designphilosophy with strong geometric similarity, significantlyimproving scale-up success rates. Consistent hydrody.namic conditions across scales (e.g., mixing efficiencyand shear distribution)minimize process variation andensure stable cell culture performance.
The bottom-mounted axial flow impeller (e.g., marine impeller) drives vertical circulation within the vessel, generating large-scale bulk flow and minimizing local dead zones and turbulence heterogeneity, thereby improvingoverall mixing perfommance. lt increases gas residencetime in the liquid phase, resulting in enhanced gas-liguid mass transfer. Compared with radial flow impellers,it provides lower shear stress, reducing the risk otdamage to shear-sensitive cells (e.g., mammaliancells). Macro-bubbles (~0.8 mm)and micro-bubbles(~150 um)support high-density cell culture while alsoenabling efficient CO₂ stripping.

Flexible Configuration
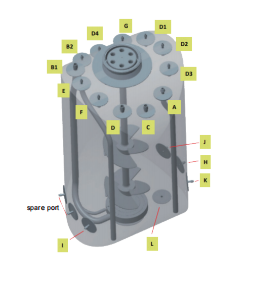
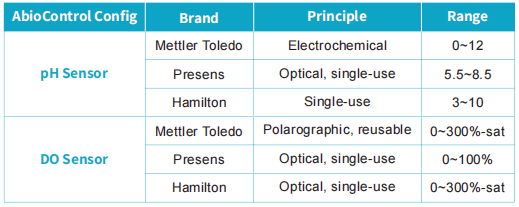
Based on Applitech's extensive experience in upstream applications, Abiobag has been further optimized ontop of standard configurations. For example, the exhaust filter is pre-integrated with the bag and gamma-irradiated as a complete assembly, eliminating the need for on-site connections andreducing contamination risk during operation. In addition, the number of ports, aswell as tubing diameter and length, can be flexibly customized according toprocess requirements. For sensors, up to three different types of pH and DOprobes are supported, covering the mainstream probe technologies currently usedin the industry.
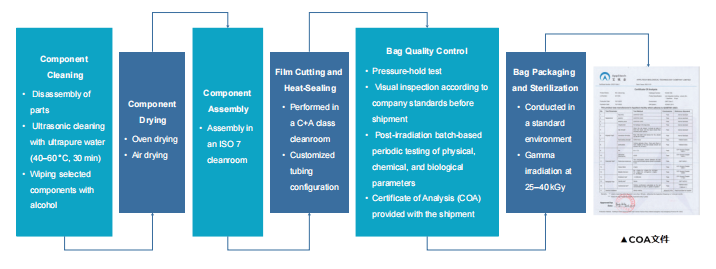
Consistent Batch Quality
AbioBag is manufactured under a comprehensive production process and quality system. incoming inspectionis performed on the film, including visual inspection, heat-seal strength testing, and endotoxin assessmentDuring production, all personnel undergo formal training and are qualified before operation. Components areassembled in an 1$0 7 cleanroom to prevent any insoluble particulates from entering the bag. After productioneach bag undergoes strict quality control, including a pressure-hold test to ensure the integrity of every bagbefore leaving the factory.