Company news
Company news
The “Invisible Engine” Behind Precision Mixing: How Single-Use Mixing Systems Redefine the Efficiency Boundaries of Biopharmaceutical Manufacturing
In the production of advanced biologics such as antibodies and cell therapies, excessive deviations in mixing accuracy can result in the loss of entire batches. The long-standing challenges of cleaning validation and limited flexibility associated with traditional stainless-steel mixing tanks are now being addressed by the technological revolution of Single-Use Mixing Systems (SUMS).
I. Industry Pain Point: The Efficiency Dilemma of Traditional Mixing Systems
A Real Case from a CDMO
“Switching products with traditional stainless-steel mixing tanks requires 72 hours of CIP/SIP validation. Significant deviations in buffer preparation led to conductivity exceeding specifications in a clinical batch, resulting in immeasurable losses.”
Pain Points of Traditional Stainless-Steel Mixing Tanks
-
Cleaning Risks: Residual carryover and cross-contamination (critical for toxic molecules)
-
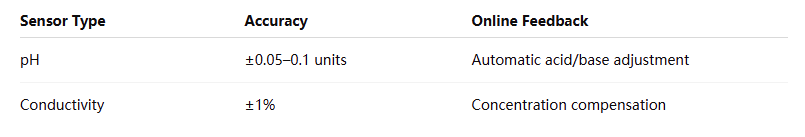
Precision Issues: pH and conductivity fluctuations > ±5% (vs. ±1% required for ultra-sensitive formulations)
-
Capacity Loss: Up to 30% of production cycles consumed by cleaning validation
-
Lack of Flexibility: Multi-product lines require repeated validation
Data Insights
According to a survey by BioProcess International, companies adopting Single-Use Mixing Systems reported a 67% reduction in batch failure rates and an 85% decrease in product changeover time.
II. Breaking Technical Barriers: The Innovative Architecture of Single-Use Mixing Systems
Dynamic Mixing Core: From Mechanical Agitation to Non-Contact Drive
-
Magnetic Coupling Mixing Technology:
-
Optimized coupling strength minimizes risk of decoupling
-
Turbulence control with CFD-optimized impeller design achieves mixing uniformity (CoV) < 5%
-
-
Magnetic Levitation Mixing Technology:
-
Floating rotor provides higher precision
-
Non-contact design reduces frictional heating from impellers
-
Low shear force, ideal for sensitive and specialized processes
-
Parameter Control: Real-time monitoring and automated regulation
Modular Consumable Design: Single-Use Fluid Pathways
-
Multilayer Co-extruded Bag:
-
Inner Layer: ULDPE (excellent biocompatibility)
-
Oxygen Barrier Layer: EVOH (reduces gas and vapor permeability)
-
-
Aseptic Connection Technology:
-
Aseptic docking technology replaces traditional tube welding
-
Operation time < 1 minute, significantly reducing contamination risk
-
III. Application Breakthroughs: Three Key Value Scenarios
High-Viscosity Solution Preparation (e.g., cellulose colloids)
-
Pain Point: Traditional mixing dead zone > 15%
-
Solution: Eccentric magnetic impeller design
-
Outcome: Achieves target mixing uniformity even under high-viscosity conditions
Multi-Product Manufacturing on Shared Production Lines (e.g., CDMO facilities)
-
Pain Point: Excessive cost and time consumed by cleaning validation
-
Solution: Complete replacement of bag and fluid path
-
Outcome: Product changeover time reduced from several days to just a few hours
IV. Proactive Prevention: Preventive Solutions for High-Frequency Failures

Applitech offers the AbioMixer desktop mixing system and single-use bottom-mounted magnetic mixing systems, integrating multiple functions including stirring, weighing, printing, pH, conductivity, and temperature control. With continuously optimized software, the systems enable precise parameter regulation, ensuring minimal deviation throughout the mixing process.

Figure 1. Applitech AbioMixer Desktop Mixing System and Single-Use Bottom-Mounted Magnetic Mixing System
When paired with the Haimore series single-use storage bags, the systems come with comprehensive and reliable validation documentation. They can be used without the need for cleaning validation, helping biopharmaceutical companies achieve faster and more efficient production.

Figure 2. Applitech Haimore Single-Use Storage and Mixing Bag
Conclusion:
Single-Use Mixing Systems have evolved from being merely an “alternative option” to becoming a core enabler of bioprocessing. Their value extends beyond eliminating cleaning validation; by providing precise and controllable fluid handling, they offer scalable, traceable, and highly compliant foundational support for cutting-edge fields such as cell and gene therapies and mRNA vaccines.
Applitech Biological Technology Co., Ltd., as a high-tech enterprise integrating design, R & D, production, sales and
service, we are committed to providing professional production and analysis equipments, single-use consumables and
comprehensive solutions for the biopharmaceutical field. Since our establishment, we have always been custom
er-centered and taken "Quality First, Service Foremost" as our business principle. Based on the optimization, scale-up
and production of bioprocess, we have been continuously improving the product lines in the biopharmaceutical field,
providing users with all-round professional solutions for biotechnology and helping them to continuously make new
breakthroughs in the biopharmaceutical field.






